Local Therapeutics Company Pioneering Nanotech

Complete the form below to unlock access to ALL audio articles.
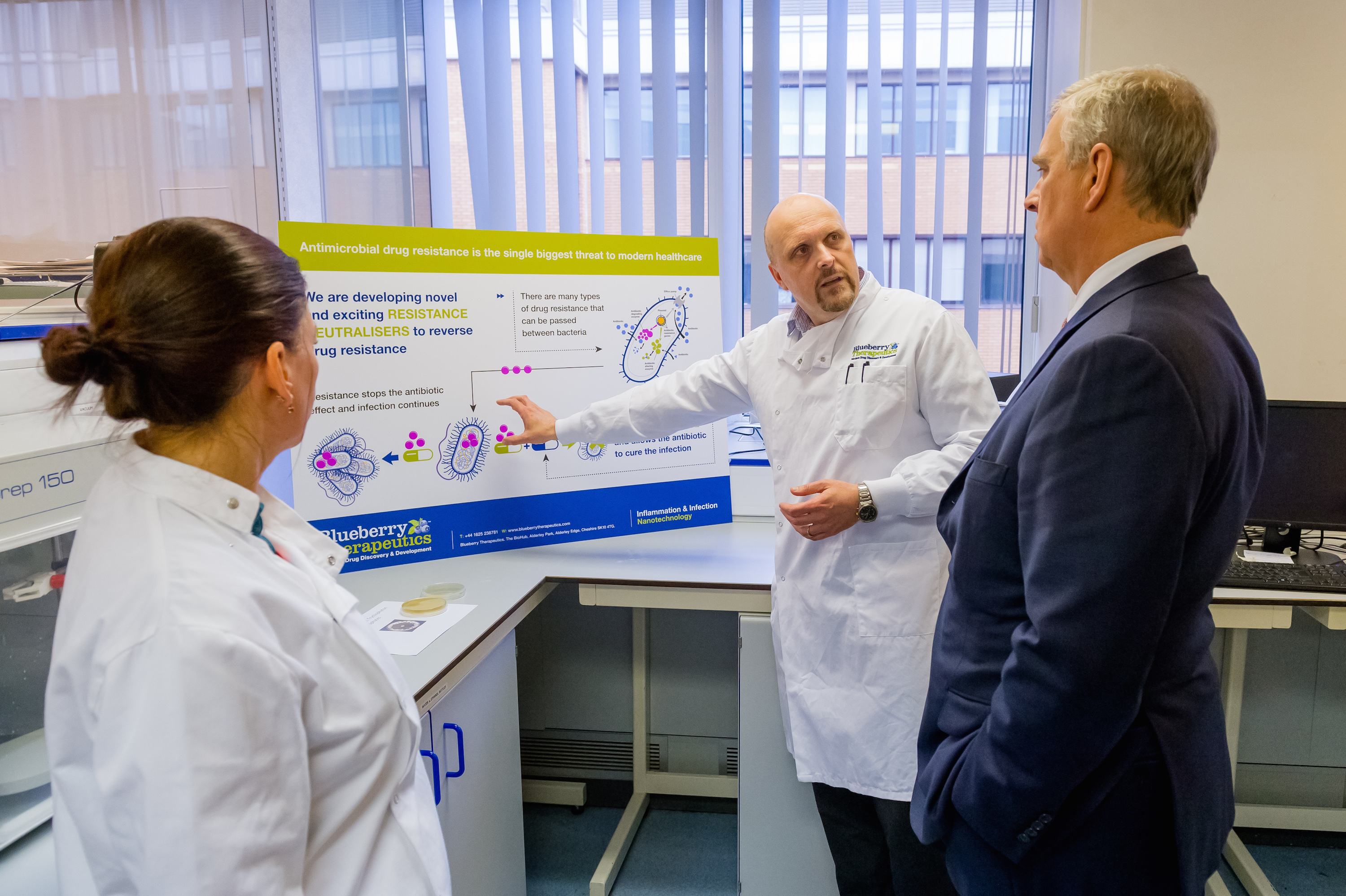
Blueberry Therapeutics Ltd, a leading pharmaceutical company focused on the major medical problems of inflammation and infection, are exploring the anti-microbial agenda through the development of new anti-fungal drugs and treatments to tackle this important unmet healthcare need. Located in the BioHub at Alderley Park, Cheshire, they recently received a visit from the Duke of York who was interested to hear about Blueberry’s goals to develop new anti-infective treatments and see first hand some of the nanoparticle formulation and characterisation work.
Nanotechnology is often hailed for its potential to advance medicine, however it is not simply working at ever smaller dimensions; rather, working at the nanoscale enables scientists to utilise the unique physical, chemical, mechanical, and optical properties of materials that naturally occur at that scale. Blueberry has unique nanopolymer delivery technology that opens up huge opportunities to rapidly develop new medicines and help find ways to treat infection and inflammation.
Blueberry’s novel nanotechnology drug delivery platform allows the company to deliver drugs into cells and tissue in an efficient way, which has the potential to facilitate the development of therapies to tackle the huge healthcare needs of antibiotic resistance in bacterial infection. As they can deliver drugs so efficiently with this platform, Blueberry can:
• Cut doses – and potential side effects – while maintaining efficacy
• Make changes inside cells to make them susceptible to drugs – with the potential to revitalise old antibiotics that don’t work
Dr Mike Davies, Co-Founder and Chief Medical Officer at Blueberry Therapeutics, commented: “Last year we secured investment that was an extremely significant stepping stone in our journey to deliver new therapies to patients suffering from serious bacterial and fungal infections. The capital will enable Blueberry to progress two anti-fungal programmes to clinical trials this year. In the short-term we are delivering a portfolio of over-the-counter products to generate revenue and thus enable the growth of Blueberry and ensure significant return to our investors.”
On the visit from HRH Duke of York, Dr Davies added: “We were delighted to be able to host The Duke and present our goals to develop new anti-infective treatments. The Duke of York saw first hand some of our nanoparticle formulation and characterisation work.”
Blueberry Therapeutics utilise a highly effective drug delivery system based on a well-described nanopolymer with over 30 years of safe human use. This system rapidly self-assembles with both small and large molecules (e.g. proteins, peptides and nucleic acids) and “packages” them into nanoparticles. These nanoparticles greatly enhance cellular delivery into both mammalian and bacterial cells.
Use of this technology allows Blueberry to develop therapies against well-validated targets in new ways. It also helps to over-come the limitations of some current therapies by improving drug solubility, cellular delivery and efficacy. Finally, the ability of the nanopolymer system to deliver protein and peptides in active confirmations into cells opens up exciting new possibilities for “intracellular biologics” capable of tackling attractive approaches to disease treatment that are currently perceived to be “undruggable” by conventional small molecule treatments, such as neutralising resistant bacteria.
Blueberry Therapeutics is committed to the development of innovative therapies to treat diseases where infection and inflammation are major factors, using cutting-edge nano polymer drug delivery technology to exploit both small molecule and biologic approaches to developing treatments.

