Test Drinking Water for Arsenic in Just One Minute
Complete the form below to unlock access to ALL audio articles.
A low-cost, easy-to-use sensor which can test drinking water for arsenic in just one minute has been developed by Imperial and UCL researchers.
Worldwide, 140 million people drink water containing unsafe levels of arsenic, according to the World Health Organisation. Short-term exposure causes skin lesions, skin cancer and damage to the cognitive development of children, while long-term exposure leads to fatal internal cancers.
In one region of Bangladesh, one of the worst-affected countries, around 20% of all deaths are attributable to arsenic poisoning.
"This sensor to detect harmful levels of water contamination will make a huge difference across developing nations, potentially saving millions of lives" commented Sam Gyimah, Universities and Science Minister.
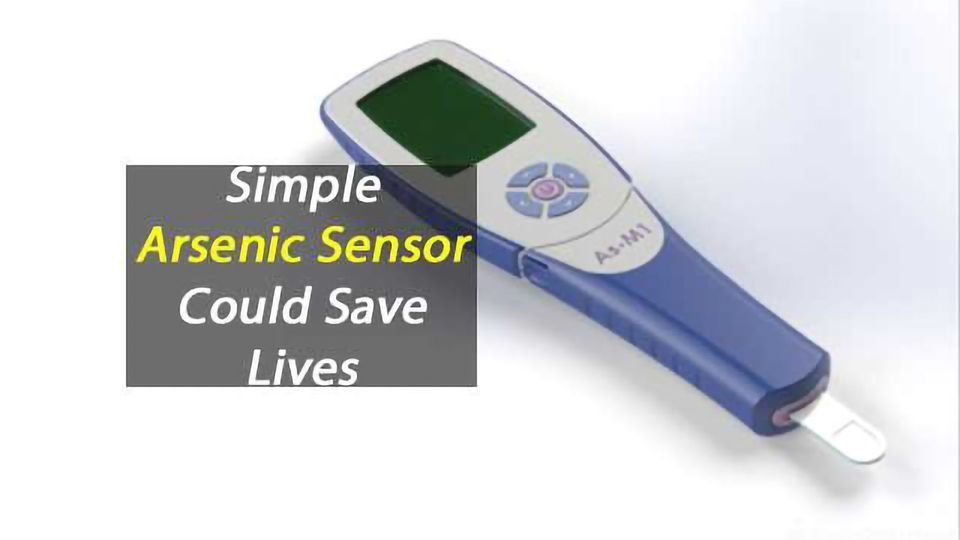
Researchers from Imperial College London and University College London (UCL) have developed a small, sensitive and accurate sensor, which produces an immediate measure of the arsenic level in water at a cost of less than $1 per test.
Currently available tests for arsenic need to either be carried out by scientists in a laboratory or using chemical test kits that produce toxic chemicals as by-products and take up to half an hour to give a result.
The new sensor, developed with BBSRC funding through the government’s Industrial Strategy, resembles the blood glucose meters used by diabetics. Once a drop of water hits the test strip, which is inserted into the sensor, it produces a digital reading of the arsenic level in one minute.
This would enable it to be used by local people in rural areas in low and middle-income countries, where the problem of arsenic poisoning is greatest and access to healthcare and technology is limited.
Creating the technology of tomorrow
Commenting on this game-changing product, Universities and Science Minister Sam Gyimah said: “This sensor to detect harmful levels of water contamination will make a huge difference across developing nations, potentially saving millions of lives.
“Too many people are exposed to dangerous levels of arsenic and this product is a clear demonstration of our Industrial Strategy in action, creating the technology of tomorrow and supporting the high-value, high skilled jobs that will make Britain fit for the future.”
The concept for the device was developed by Dr Joanne Santini from UCL and Professor Tony Cass from the Department of Chemistry at Imperial, whose earlier work led to the development of the first electronic blood glucose monitor.
Dr Santini had the idea of developing a sensor to detect arsenic in water when she discovered a microscopic organism that eats arsenic. She realised that an enzyme produced by this organism could be used to measure arsenic levels.
Bio Nano Consulting, a spin-out from Imperial and UCL, has patented the sensor design and, using a Smart Award from Innovate UK, created a prototype which it can produce in batches of up to 100.
Success in the field
Professor Cass said: “It’s been a real journey to take an idea, conceived of at a London Centre for Nanotechnology research day, through proof of concept research, then into prototyping and scale up and now to field trials in Bangladesh. Now the technology has been spun out we can look forward to scale up of manufacture and ultimately deployment.”
Dr David Sarphie, CEO of Bio Nano Consulting, said: “Arsenic contamination of water is a hugely important issue, and the technology hasn’t existed to enable it to be addressed properly. We feel that a user-friendly, cheap and rapid test could be extremely beneficial in terms of revolutionising how the problem might be addressed.
“We were doing some early-stage field trials a few months ago in Bangladesh and a lot of the villagers were actually pleading with us to come and measure their well, because they had no idea how much arsenic was in their water.”
The researchers expect it to be on the market within three years.
This article has been republished from materials provided by Imperial College London. Note: material may have been edited for length and content. For further information, please contact the cited source.

