Protein Biotherapeutics: Mass Spectrometry Analysis

Complete the form below to unlock access to ALL audio articles.
Mass spectrometry techniques
Increased demand for biopharmaceutical companies to identify new drugs and to uncover novel therapeutic targets has meant a greater interest in exploring the therapeutic potential of less abundant cellular proteins. This shift in focus has been aided by the significant advancements within the field of proteomics, specifically by the improvement of mass spectrometry (MS).
Its ability to scrutinize physical and chemical protein stability as well as the pharmacokinetic properties of a protein makes it a practical, flexible tool for use during biopharmaceutical development. A modification to either the primary, secondary, tertiary, or quaternary structure of a protein can have a huge impact on the safety and overall effectiveness of a biotherapeutic, demonstrating the importance of robust, versatile bioanalysis methods.1
MS has evolved as a prevailing technique for both the characterization of biotherapeutics and identification of protein variants and post-translational modifications. The versatility of MS technologies means that it can take many different forms dependent on your analysis needs.
The most frequently used soft ionization techniques for protein mass spectrometry include liquid chromatography/electrospray ionization-MS (LC-ESI-MS/MS) and matrix assisted laser desorption ionization-time of flight (MALDI-TOF) MS.2 Soft ionization allows you to sustain a low internal energy in the analyte throughout the entire process, meaning that the primary polypeptide backbone remains whole.1 The type of MS workflow used for primary structural analysis of biotherapeutics will depend on the nature of the protein and its modifications (Figure 1).1

Figure 1: An overview of ‘bottom-up’- and ‘top-down’ MS analysis workflows, used to
determine the structural characterization of peptides and biotherapeutics. ‘Bottom-up’
approaches involve peptide digestion, and analysis using either MS (for peptide
mapping/peptide mass finger printing) or tandem MS. ‘Top-down’ approaches
include intact mass analysis and gas-phase fragmentation of the protein by tandem
MS. Credit: Leurs et al.
Coupling ESI-MS2 with LC allows for automated separation and processing making it the preferred MS technique for advanced ‘bottom-up’ analyses. MALDI-TOF is suited for analysis throughout biopharmaceutical development, its uncomplicated sample preparation and straight-forward data analysis make it a popular choice for peptide mapping.3
Enabling the Use of Mass Spectrometry Throughout Biopharmaceutical Development [White Paper]

This Characterization Case Study takes an inside look at a comparability study of different manufacturers lots of the biotherapeutic, trastuzumab, analyzed at the intact protein, subunit, and peptide level. You will learn how the X500B QTOF System and BioPharmaView™ Software simplifies standardized characterization analyses, giving you more complete comparability answers faster and easier.
"Mass spec users have been asking for systems that are purpose built for their application, and SCIEX has listened." Farzana Azam, Senior Director, Pharmaceutical/CRO Business at SCIEX
DOWNLOAD WHITE PAPERBioanalysis of proteins by mass spectrometry
“The number one benefit is that MS provides direct measurement of the analyte, rather than relying on indirect signals that come from one or more immune interactions.” Ian Moore, Global Technical Marketing Manager, Pharma Quant and MetID, SCIEX
More traditional techniques, including ligand binding assays (LBAs) have routinely been used for the quantification of proteins in serum. However, LBAs are somewhat impractical when it comes to large scale bioanalysis due to their expense and difficultly in their translation to a high-throughput environment, although advancement in technology and robotics is making this a more feasible approach.3 MS still dominates as the analytical tool of choice due to its superior selectivity between structurally similar proteins, its compliance to a high-throughput setting, general robustness and reduced demand for specific reagents.3
“MS platforms are universally applicable: all proteins and peptides can be evaluated by LC-MS/MS, whereas ligand binding assays require specific antibodies for each analyte. As a result, LC-MS/MS assays have a much wider linear dynamic range (LDR) of 4-5, and can provide selective data even when working with samples containing a complex background of endogenous proteins. LC-MS/MS also offers benefits of speed: methods can be developed and validated in a shorter period of time, and for analysing multiple targets at once.” Ian Moore, Global Technical Marketing Manager, Pharma Quant and MetID, SCIEX
Investigating ADME, pharmacokinetics, and metabolite ID using mass spectrometry
“It is important to study [ADME toxicology, pharmacokinetics, and metabolites] to make effective and safe drugs. MS is the best tool for all three studies, due to its selectivity, specificity, throughput and sensitivity.” Ian Moore, Global Technical Marketing Manager, Pharma Quant and MetID, SCIEX
The distribution and elimination of biotherapeutics is a major challenge, and attention must be directed toward determining target mediated clearance, immunogenicity, and metabolic stability. The most informative studies are usually performed in vivo and involve tracking the drug/metabolite’s journey throughout the body, using MS techniques.
“Performing MetID studies is an important part of understanding overall ADME. Due to the risk of toxicity from metabolites, MS analysis is a powerful tool for advanced metabolism studies where it is important to try and identify all potential metabolites/catabolites, to ensure safety of a therapy. Advanced MS technology, such as SWATH Acquisition, allows us to be more confident that we can detect even low-level compounds that may be harmful.” Ian Moore, Global Technical Marketing Manager, Pharma Quant and MetID, SCIEX
Quantitative mass spectrometric assays have been advancing over the last ten years and will continue to be one of the most powerful methods for performing toxicological analysis of biotherapeutics.3
Glycosylation analysis by mass spectrometry
Post translational modifications (PTMs) can influence a protein’s activity and structure, cause aggregation, ultimately affecting immunogenicity and other pharmacological properties. It is therefore important to identify and understand the impact these modifications have during biopharmaceutical manufacture to ensure any impact can be adequately controlled.4 Fortunately, MS is a valuable tool for the detection and analysis of PTM’s. A key benefit is its reproducibility, accuracy, and flexibility; MS can be coupled to other techniques, such as LC or capillary electrophoresis.5 It can be used both early in the drug development pipeline, and later on, during manufacturing and quality control stages, prior to final release.
As well as being the most complex, glycosylation is also the most common PTM of proteins. More specifically, >60% of biotherapeutics are glycosylated.6 It is important to monitor glycosylation throughout the entire production process as this can be influenced by a number of different factors including the type of expression system used, cell culture protocols and downstream purification techniques.7
When you consider biotherapeutics, there are two main types or glycosylation to be aware of: N-linked, whereby the glycan is attached to an asparagine; O-linked, whereby the glycan is attached to either a serine or threonine residue (Figure 2).
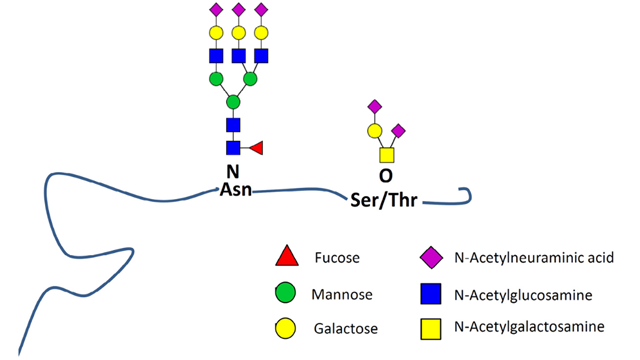
Figure 2: Model glycoprotein showing both N-linked and O-linked glycosylation. Credit: Dotz et al.
“Glycosylation was identified as being one of the features on biotherapeutics that is key for safety. This originates from a time when biotherapeutics were grown in murine cells and mouse glycans were found to be immunogenic. These days, monitoring and manipulating a biotherapeutic’s glycosylation is a way to manipulate the pharmacology, in order to improve efficacy and safety.” Sibylle Heidelberger, Technical Marketing Manager Biologics, SCIEX
To ensure efficacy and safety, biotherapeutics should display human-compatible and consistent glycosylation and as such it is a regulatory obligation to characterise N-glycosylation.7 Regulatory agencies including the US Food and Drug Administration and European Medicines Agency, have produced guidelines on the requirement to assess the role glycosylation plays in modulating immunogenicity of biopharmaceutical protein products.
“Glycosylation can be analyzed using three techniques: maintaining the intact protein form to monitor the distribution and levels of glycosylation; digestion of the protein into peptides; and maintaining the location and glycosylations’ intact and release form of the glycans to characterise and quantify the glycan chains. Different types of MS can be used for these analyses, and they all use high resolution mass spectrometers such as Time-of-Flight (TOF) technology. The resolution and mass accuracy means that they are capable of analyzing the large intact glycosylated protein precisely as well as analysing released glycans and glycopeptides. Such high-resolution instruments are normally also capable of doing MS/MS, which gently induces specific fragmentation of glycans and glycopeptides, allowing for confirmation of the glycan sequence, as well as the location and sequence of the peptide to which they are attached.” Sibylle Heidelberger, Technical Marketing Manager Biologics, SCIEX
Mass spectrometry and industry progression
The proactive approach companies are taking to continually improve the sensitivity, resolution, mass accuracy and range of MS technologies coupled with the flexibility to apply these to all levels of the biopharmaceutical process – from early discovery to release of approved product, makes MS an attractive and valuable tool for the analysis and development of biotherapeutics.
“Adopting novel MS techniques is important to biopharma as it is to any other manufacturing. The key to new techniques is that they should facilitate and speed up the analytical process. The result is to allow the manufacturer to release a product faster while maintaining the safety of the product.” Sibylle Heidelberger, Technical Marketing Manager Biologics, SCIEX
The FDA has introduced a quality by design (QbD) approach8 for monitoring biotherapeutics which aims to improve the efficiency and design of product during the manufacturing process.
“QbD has led to the identification of Product Quality Attributes (PQA) specific to each biotherapeutic, that can be product- or process-derived. These are monitored since they have the ability to affect the safety or efficacy of the drug. Previously these attributes would have been monitored individually through multiple techniques such as ELISA, CEX or HILIC, however the move towards LC-MS peptide mapping for identifying and confirming sequence and post-translational modifications has also led to the development of a single Multi-Attribute Method (MAM) to track and monitor key PQAs.” Sibylle Heidelberger, Technical Marketing Manager Biologics, SCIEX
Moving towards a single method, a single assay, to monitor as many PQAs as possible maximises efficiency. MS has a huge part to play in this field with advancements in MS technologies set to positively influence the discovery, development and approval of novel biotherapeutics in the future.


