Sugar Molecules as Targets in Cancer Therapy
Complete the form below to unlock access to ALL audio articles.
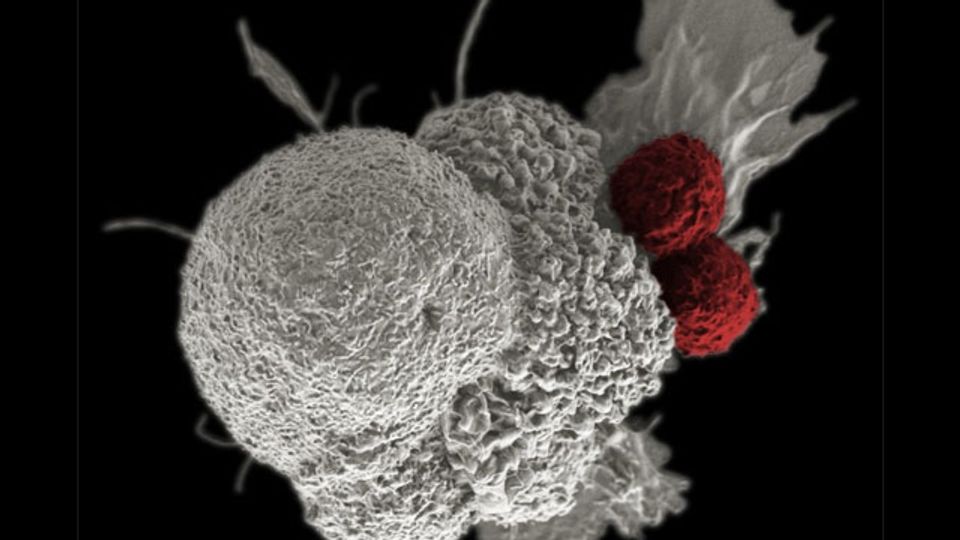
Cancer cells use sugar molecules on their surface to disable attacks by the body’s immune system. Researchers at the University of Basel now report on how this mechanism can be neutralized.
The immune system is actually extremely well equipped to get rid of abnormal cells. As a safety mechanism, special features are built into healthy cells so that the immune system recognizes them, thus preventing a mistaken attack. However, cancer cells sneakily manipulate these safety mechanisms in such a way that the immune system leaves them alone.
Over the past few years, immunotherapies have revolutionized cancer treatment. These include therapies that prevent cancer cells from inhibiting the immune response. This involves blocking what are known as “immune checkpoints” using artificially produced proteins, which allows the immune cells to successfully attack the cancer cells.
“With many tumors, however, there have only been modest levels of success. That’s why we’re looking for new approaches to engage anti-tumor immune responses more efficiently,” explains Professor Heinz Läubli from the Department of Biomedicine at the University of Basel and the University Hospital Basel. In the specialist journal Science Translational Medicine, his team, together with that of recent Nobel laureate Professor Carolyn Bertozzi from Stanford University, reports on a promising new approach. By altering sugar molecules on the surface of cancer cells in mice, the researchers were able to produce a significant increase in anti-tumor immune response.
How immune cells turn traitor
Their focus is on sugar molecules on the surface of the cancer cells, as well as on the cells in their immediate vicinity. These particular sugars, which contain sialic acid, also occur on healthy cells, and are important for cell-to-cell communication. However, tumors boost the proportion of these sugars on their surface.
Certain immune cells called macrophages recognize these sialic acid sugars and inadvertently turn traitor: they give other nearby immune cells the impression that all is well. The research team’s experiments on mice have now been able to demonstrate that the sialic acid sugars can be removed, or at least very much reduced, with the help of an enzyme. This means that the macrophages no longer prevent the tumor coming under immunological attack.
A target structure for new therapies
More precise analyses have enabled the researchers to identify in mice exactly which receptor it is on the macrophages that recognizes the sialic acid sugars. If the equivalent receptor could be identified in humans, then that could be another interesting target in the bid to tackle cancer cells with the aid of the patient’s own immune system.
“The combination of our approach with the immune checkpoint blockade methods that have already been established meant we could really put the brakes on tumor growth in the laboratory mice,” says Professor Läubli. As a next step, the researchers aim to seek ways of removing the sialic acid sugars from the tumor and its surroundings in as targeted a manner as possible, in order to avoid disrupting the function of healthy cells and eliminate side effects.
Reference: Stanczak MA, Rodrigues Mantuano N, Kirchhammer N, et al. Targeting cancer glycosylation repolarizes tumor-associated macrophages allowing effective immune checkpoint blockade. Sci Transl Med. 2022;14(669):eabj1270. doi: 10.1126/scitranslmed.abj1270
This article has been republished from the following materials. Note: material may have been edited for length and content. For further information, please contact the cited source.

