Natural Immunity to Malaria Offers Direction for Future Therapies
Complete the form below to unlock access to ALL audio articles.
WEHI researchers have identified how natural human antibodies can block malaria parasites from entering red blood cells, potentially indicating how new protective therapies could be developed against this globally significant disease.
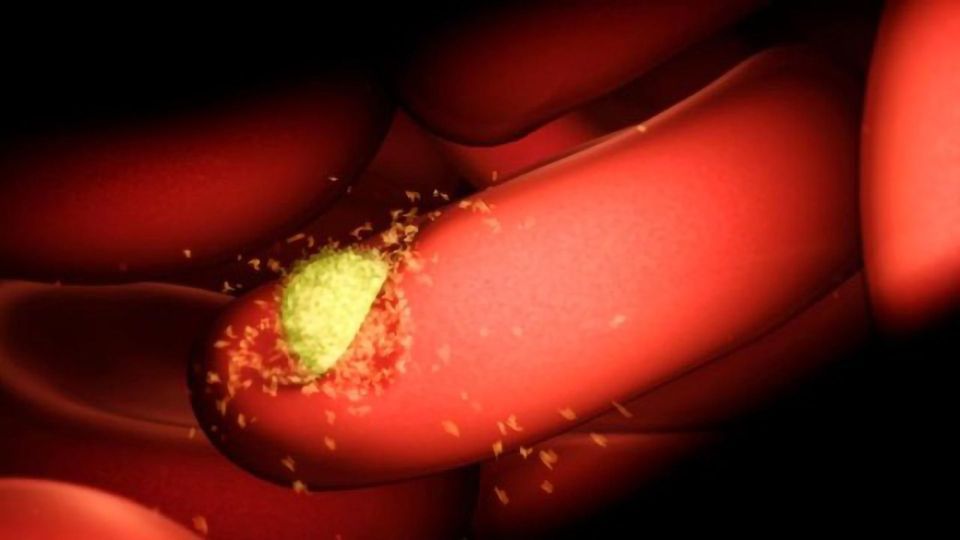
The research provides greater insight into how antibodies block the entry of Plasmodium vivax malaria parasites into young red blood cells called reticulocytes. It builds on an earlier discovery that the P. vivax latches onto the transferrin receptor 1 (TfR1) to enter cells.
The research, led by Associate Professor Wai-Hong Tham and PhD student Li-Jin Chan from WEHI, alongside Professor Christopher King from Case Western University, US, was published in Nature Communications.
Shedding light on pathogen-blocking antibodies
Plasmodium vivax is the most widespread malaria parasite in the world, and the predominant cause of malaria in the vast majority of countries outside Africa. It is also the main parasite responsible for recurrent malaria infections.
The malaria parasite is a complex single-celled organism, with diverse proteins that help it to invade red blood cells, reproduce and spread. Adhesins on the surface of the parasite are key-like proteins that 'unlock' cells, allowing the parasite to enter.
Previous research studies in Papua New Guinea, Thailand and Brazil showed antibodies against P. vivax adhesins were correlated with protection against infection and disease, Associate Professor Tham said.
"We wanted to understand how these human antibodies in natural infection block the parasite from getting in. By extracting and examining antibodies from people who have had P. vivax infections, we identified the different ways human antibodies against P. vivax work. One of these ways, is by stopping the parasite adhesins from getting too close to the reticulocyte membrane, denying the parasite entry," she said.
This discovery opens the door to potentially preventing not only P. vivax malaria, but also P. falciparum malaria, another significant cause of deaths globally.
"Although this was a vivax study, we believe the implications are that a broadly neutralising antibody could be created to target both P. vivax and P. falciparum malaria infections," Associate Professor Tham said.
Improving detection of relapsing malaria
WEHI Professor Ivo Mueller said beyond understanding how antibodies can block infection, there was also a crucial need to understand the development of immunity and how this could be used to detect P. vivax infections in endemic populations.
"We are currently using this information to develop diagnostic tests that will be used in the field to identify and treat people with hidden vivax infection in their livers and spleens. This is a key step towards eliminating malaria, by preventing silently infected people reinfecting their communities," he said.
This work was made possible with funding from the National Health and Medical Research Council, the European Research Council and the Victorian Government.
Reference
Chan L-J, Gandhirajan A, Carias LL, et al. Naturally acquired blocking human monoclonal antibodies to Plasmodium vivax reticulocyte binding protein 2b. Nature Comms. 2021;12(1):1538. doi:10.1038/s41467-021-21811-2
This article has been republished from the following materials. Note: material may have been edited for length and content. For further information, please contact the cited source.

