The Scalable Ergonomic Mobius® Bioreactor Platform

Want to listen to this article for FREE?
Complete the form below to unlock access to ALL audio articles.
Read time: 2 minutes
EMD Millipore recently introduced the Mobius® 2000 liter single-use bioreactor. The 2000 liter bioreactor joins the Mobius® stirred- tank bioreactor portfolio (from 3 to 2000 liters) that provides the ultimate in flexibility, scalability, and convenience.
To learn more about this exciting addition to the Mobius® stirred- tank bioreactor portfolio (from 3 to 2000 liters) and the benefits it provides researchers, we spoke to Janice Lloyd Simler, PhD Global Product Manager, Bioreactor and Clarification Systems, EMD Millipore.
AB: The Mobius® range already offered 3, 50 and 200 L bioreactors; was it always the intention to expand the range to include a 2000 L offering? Did the features of these bioreactors readily enable scale up to this latest version?
JS: The intention was to offer a 2000 L bioreactor to complete the product portfolio and provide a scalable solution from 3L through 2000 L. Many of the design features in the 50 and 200 L bioreactors are also present in the 2000 L bioreactor such as the impeller design and placement, the low turn down ratio, the presence of a baffle and the sparger design. The common design features along with the characterization of several key engineering parameters of the 50 and 200 L bioreactors enabled scale-up to the larger bioreactor.
AB: Can you share some data that demonstrates the scalability of the platform?
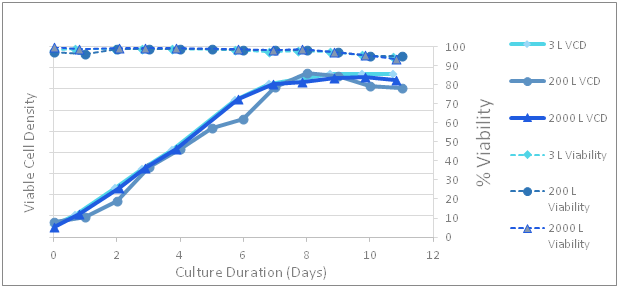
JS: Several parameters can be used to scale-up a cell culture process including geometric similarity, the impeller design tip speed, power input per volume and oxygen mass transfer coefficient (kLa). As shown in Table 1, we have maintained geometric similarity across the platform for several important ratios. However, keeping these ratios constant does not necessarily guarantee scalable cell culture performance. Therefore, we have characterized some key engineering parameters including determining the mass transfer coefficient (kLa) values, tip speed and mixing. The first cell culture run in the Mobius® 2000 L bioreactor was performed at the Merck KGaA, Darmstadt, Germany BioDevelopment facility in Martillac, France. As shown in Figure 1, there was comparable cell growth and viability in the 3 L, 200 L and 2000 L bioreactor.
Table 1.
AB: Can you tell me more about the bottom loading drawer and the impact this has on the Flexware® assembly?
JS: Single-use assemblies greater than 1000 L can be cumbersome, especially in a manufacturing environment where space is limited and there are concerns that excessive handling can damage the assemblies. The Mobius® 2000 L bioreactor was designed to enable easy Flexware® Assembly installation and removal. The location of the impeller and the front plate in the Flexware® Assembly helps to orient the direction of the bag during installation resulting in correct placement in the vessel drawer on the first try. Further, the Mobius® 2000 L Flexware® Assembly features EZ fold technology, which allows for hands-free inflation. These design elements reduce the need for additional equipment for single-use assembly installation, ensure ease-of-use, consistent single-use installation and safety for the operator.
AB: The 2000 L bioreactor has a novel X baffle design, do you have any data that demonstrates its effectiveness?
JS: During development of the 2000 L bioreactor, mixing videos were recorded in the presence and absence of the novel X-baffle. As can be seen in the video, not only does the presence of the X-baffle enable fast mixing but also homogeneous mixing. This is critical to prevent zones within the bioreactor that are not completely mixed resulting in areas of high or low pH, oxygen or nutrients which can negatively impact cell growth and viability.

To learn more about this exciting addition to the Mobius® stirred- tank bioreactor portfolio (from 3 to 2000 liters) and the benefits it provides researchers, we spoke to Janice Lloyd Simler, PhD Global Product Manager, Bioreactor and Clarification Systems, EMD Millipore.
AB: The Mobius® range already offered 3, 50 and 200 L bioreactors; was it always the intention to expand the range to include a 2000 L offering? Did the features of these bioreactors readily enable scale up to this latest version?
JS: The intention was to offer a 2000 L bioreactor to complete the product portfolio and provide a scalable solution from 3L through 2000 L. Many of the design features in the 50 and 200 L bioreactors are also present in the 2000 L bioreactor such as the impeller design and placement, the low turn down ratio, the presence of a baffle and the sparger design. The common design features along with the characterization of several key engineering parameters of the 50 and 200 L bioreactors enabled scale-up to the larger bioreactor.
AB: Can you share some data that demonstrates the scalability of the platform?
JS: Several parameters can be used to scale-up a cell culture process including geometric similarity, the impeller design tip speed, power input per volume and oxygen mass transfer coefficient (kLa). As shown in Table 1, we have maintained geometric similarity across the platform for several important ratios. However, keeping these ratios constant does not necessarily guarantee scalable cell culture performance. Therefore, we have characterized some key engineering parameters including determining the mass transfer coefficient (kLa) values, tip speed and mixing. The first cell culture run in the Mobius® 2000 L bioreactor was performed at the Merck KGaA, Darmstadt, Germany BioDevelopment facility in Martillac, France. As shown in Figure 1, there was comparable cell growth and viability in the 3 L, 200 L and 2000 L bioreactor.
Table 1.

Figure 1.

AB: Can you tell me more about the bottom loading drawer and the impact this has on the Flexware® assembly?
JS: Single-use assemblies greater than 1000 L can be cumbersome, especially in a manufacturing environment where space is limited and there are concerns that excessive handling can damage the assemblies. The Mobius® 2000 L bioreactor was designed to enable easy Flexware® Assembly installation and removal. The location of the impeller and the front plate in the Flexware® Assembly helps to orient the direction of the bag during installation resulting in correct placement in the vessel drawer on the first try. Further, the Mobius® 2000 L Flexware® Assembly features EZ fold technology, which allows for hands-free inflation. These design elements reduce the need for additional equipment for single-use assembly installation, ensure ease-of-use, consistent single-use installation and safety for the operator.
AB: The 2000 L bioreactor has a novel X baffle design, do you have any data that demonstrates its effectiveness?
JS: During development of the 2000 L bioreactor, mixing videos were recorded in the presence and absence of the novel X-baffle. As can be seen in the video, not only does the presence of the X-baffle enable fast mixing but also homogeneous mixing. This is critical to prevent zones within the bioreactor that are not completely mixed resulting in areas of high or low pH, oxygen or nutrients which can negatively impact cell growth and viability.

Janice was speaking to Ashley Board, Managing Editor for Technology Networks.

