Treatment Options for Chronic Parvovirus Viremia in Pediatric Heart Transplant Patients in a Tertiary Care Center
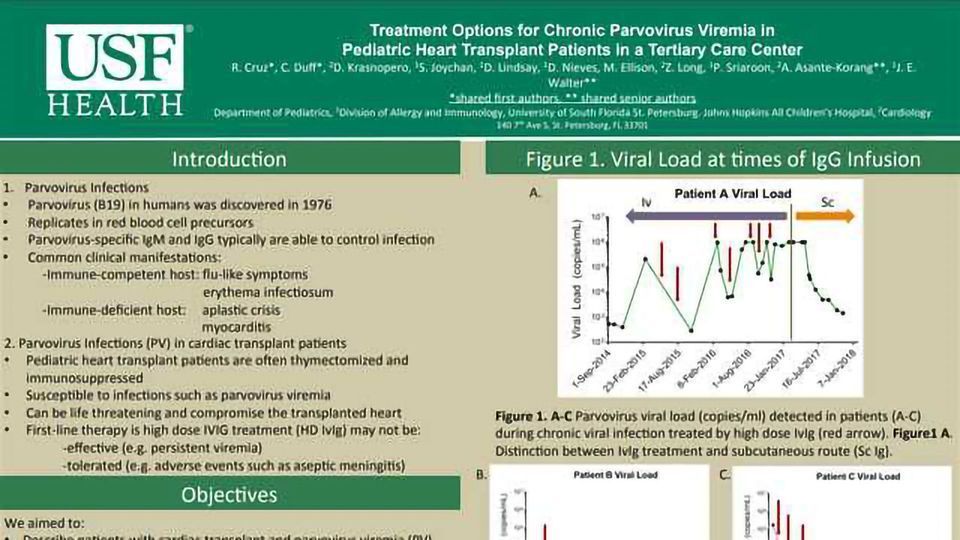
OBJECTIVE: Parvovirus viremia may occur in pediatric heart transplant patients with secondary T cell lymphopenia and history of thymectomy. While the treatment option for these patients is high dose intravenous immunoglobulin (HDIvIg), it may not be well tolerated in all patients. We present treatment outcomes in three cardiac transplant patients with parvovirus viremia.
RESULTS: Of the three patients, two responded well to treatment; however, one patient’s viral load remained at over one million copies and developed severe side effects of aseptic meningitis with HDIvIg. Compared to the other two cases, this patient had severe reduction of T cell count that likely contributed to the persistence of high viral load. In order to improve this patient’s quality of life as well as reduce the healthcare costs, a new treatment option was explored. Facilitated subcutaneous immunoglobulin treatment was initiated and resulted in a dramatic decrease in parvovirus viral loads with no adverse effects.
CONCLUSION: Pediatric cardiac transplant patients with severely reduced T cell counts may be at risk of developing chronic parvovirus viremia with high viral load. Although most patients respond well to HDIvIg, subcutaneous route may serve as an alternative to treatment refractory cases.




