Virtual Embryo Allows Single-cell Studies in Unprecedented Detail

Complete the form below to unlock access to ALL audio articles.
Researchers from EMBL Heidelberg and from the University of Padua School of Medicine have created the first complete description of early embryo development, accounting for every single cell in the embryo. This ‘virtual embryo’ will help to answer how the different cell types in an organism can originate from a single egg cell. The results are published on 20 April in the journal Cell.
“How are the many different cell types in the body generated during embryonic development from an egg, which is only a single cell? This is one of the most fundamental questions in biology,” explains Dr. Pierre Neveu, group leader at EMBL Heidelberg, setting out the rationale behind the research he and his group have performed in collaboration with the group of Dr. Lars Hufnagel.
While answering this question is essential to understand how multicellular organisms form, studying the developmental mechanisms driving this cellular diversification at the single-cell, genome-wide, and whole-embryo level is a challenging task. “So far we have lacked a comprehensive understanding of the gene expression programmes. These instruct individual cells to form the different cell types necessary to build an embryo,” explains Dr. Hanna Sladitschek, first author of the study – a former postdoc at EMBL Heidelberg and now at the University of Padua School of Medicine. Despite recent advances in the field, a complete representation of embryonic development, accounting for every single cell in space and time, has not been achieved until now.
The EMBL researchers were able to solve this problem by constructing a ‘virtual embryo’ of Phallusia mammillata – a type of marine organism known as a sea squirt, which is found in the Mediterranean Sea and the Atlantic Ocean. This species was picked as a model system because it is related to vertebrates and each individual has the same number of cells, making it easier to combine observations from many specimens.
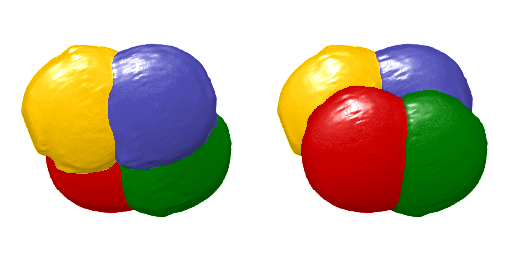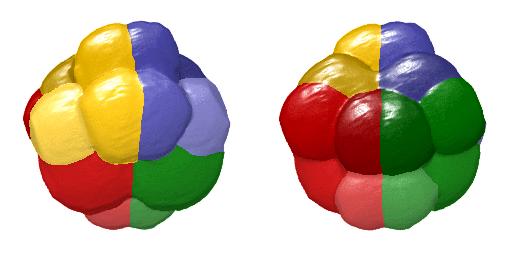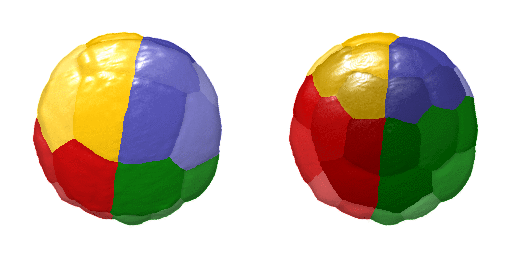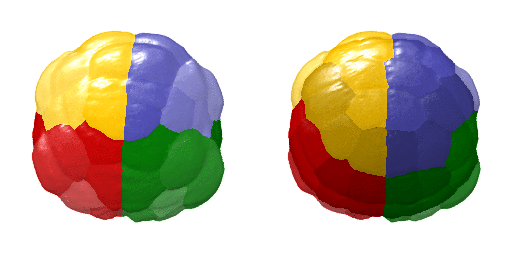
Animation showing a 4D visualisation of single-cell expression patterns. Credit: Hanna Sladitschek/EMBL
This virtual embryo describes the gene expression and morphology of every single cell of an embryo at every cell division in the early stages of development – showing the evolution from a single cell to the 64-cell stage. After these first seven cell divisions, the fates of the future nerve cord, brain, germ cells, blood cell precursors, and muscles are already specified. This makes it the first full description of early development accounting for every single cell in an embryo. It describes both gene expression – how a cell’s genetic information is expressed and appears – and spatial position. To generate this comprehensive atlas, the researchers combined high-resolution single-cell transcriptomics and light-sheet imaging.
“Our model shows that it is possible to know the location and history of an individual cell by analysing its gene expression,” says Neveu. “In addition, we find that while the regulation of gene expression is very precise within an embryo, differences in developmental timing explain the observed variation between individual embryos.”
Gene expression is generally thought to be a noisy process – in other words, one that shows an element of randomness – yet the new results show that it is remarkably reproducible and coordinated across cells in a given embryo. “How is such coordination achieved? How does the embryo coordinate between the two mirror-symmetric embryo halves?” says Neveu, highlighting some of the new questions that the scientists would like to answer.
“Our studies represent a leap forward in the emerging field of developmental genomics,” says Hufnagel. “Now that we have worked with an organism with a small number of cells, it will of course be very interesting to extend our work to mammals, which have many more cells!”
Reference: Ogai, Y., Matsumura, Y., & Hoshino, Y. (2020). Changing structures of B2B networks in the Japanese textile and apparel industry. Fashion and Textiles, 7(1), 5. https://doi.org/10.1186/s40691-019-0198-9
This article has been republished from the following materials. Note: material may have been edited for length and content. For further information, please contact the cited source.

