Complete the form below to unlock access to ALL audio articles.
Few areas in psychology have attracted as much interest as memory. Memory’s absence, amnesia, has profound implications for brain function and has been recognized as a neurological disorder for centuries. Amnesia involves the loss of declarative memory, which refers to the part of our memory that can be consciously recalled. It can be broken into two parts: episodic and semantic memory, referring to conscious recollection of autobiographical events and memory for factual knowledge, respectively. The neural underpinnings of amnesia remain a contentious and hotly debated topic. This article will examine these debates and the remaining challenges involved in understanding the neurobiological basis of amnesia.
The models
Theories regarding the neural basis of memory fall into two main camps. Associative models argue that regions within an area of the brain known as the medial temporal lobe memory system (MTLMS), consisting of the hippocampal, perirhinal, parahippocampal and entorhinal cortices, process declarative memory1. Researchers arrived at this theory after a series of studies indicated that human patients with damage to the hippocampus and surrounding areas often possess impairments in declarative memory.
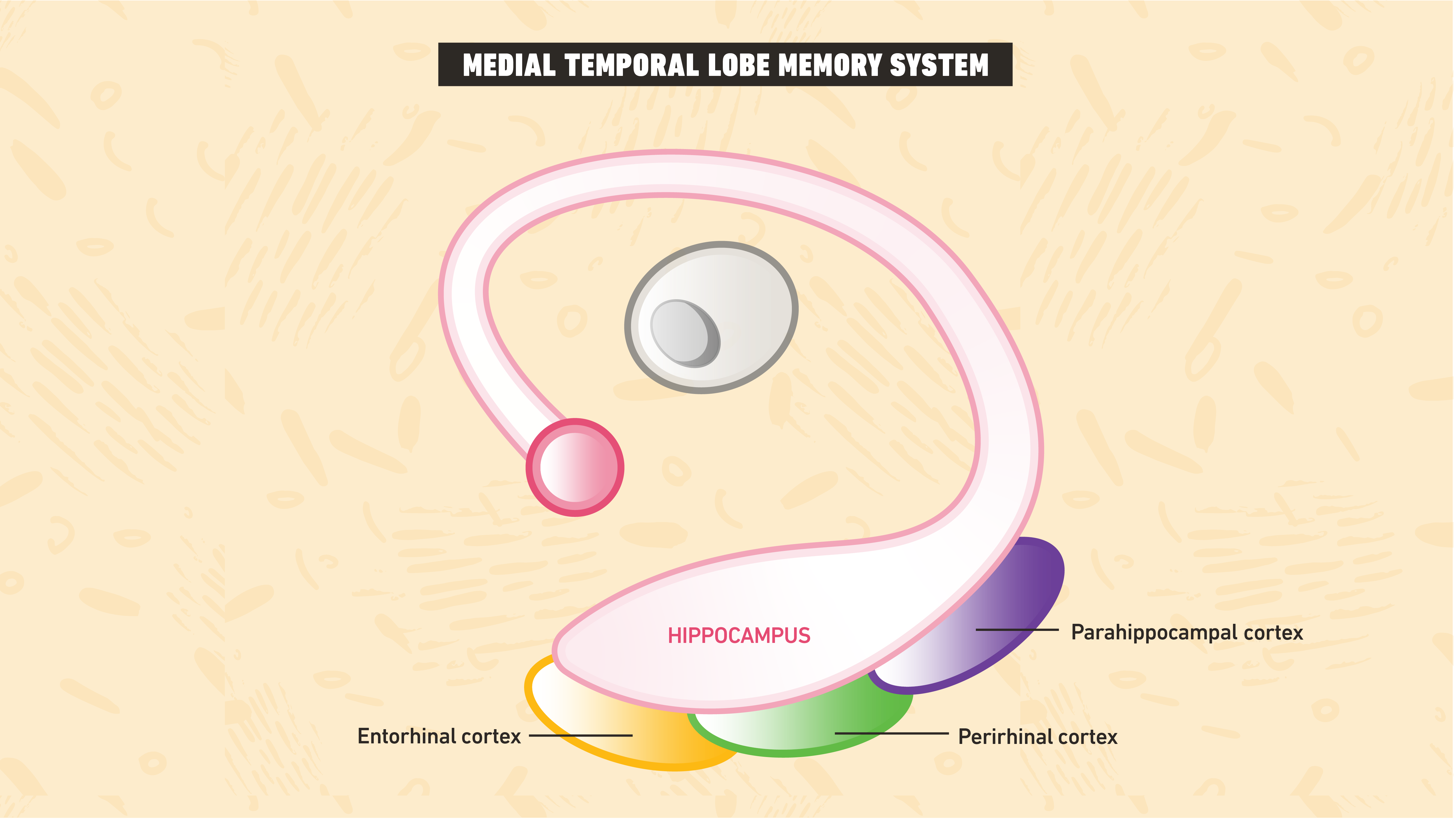
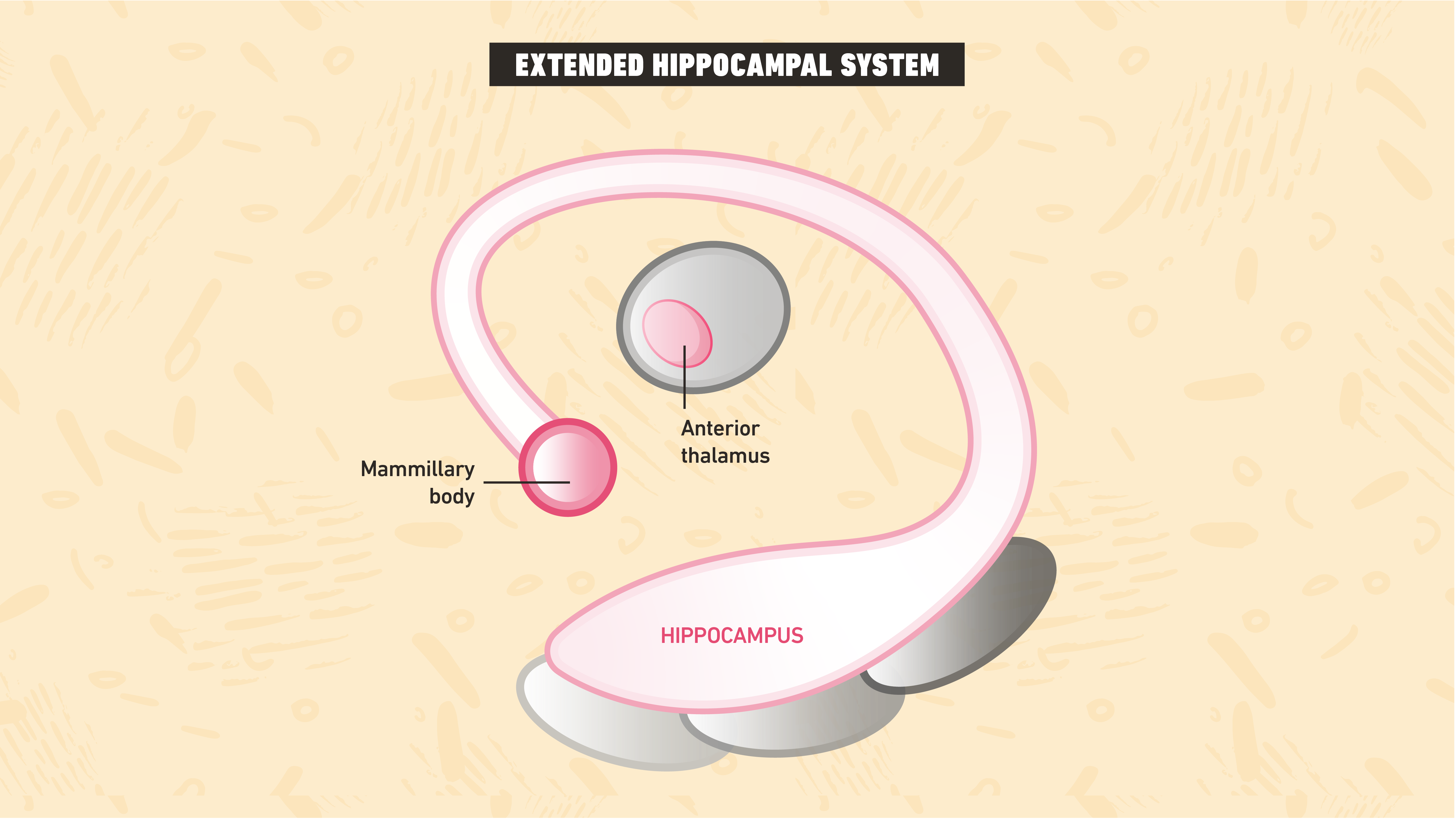
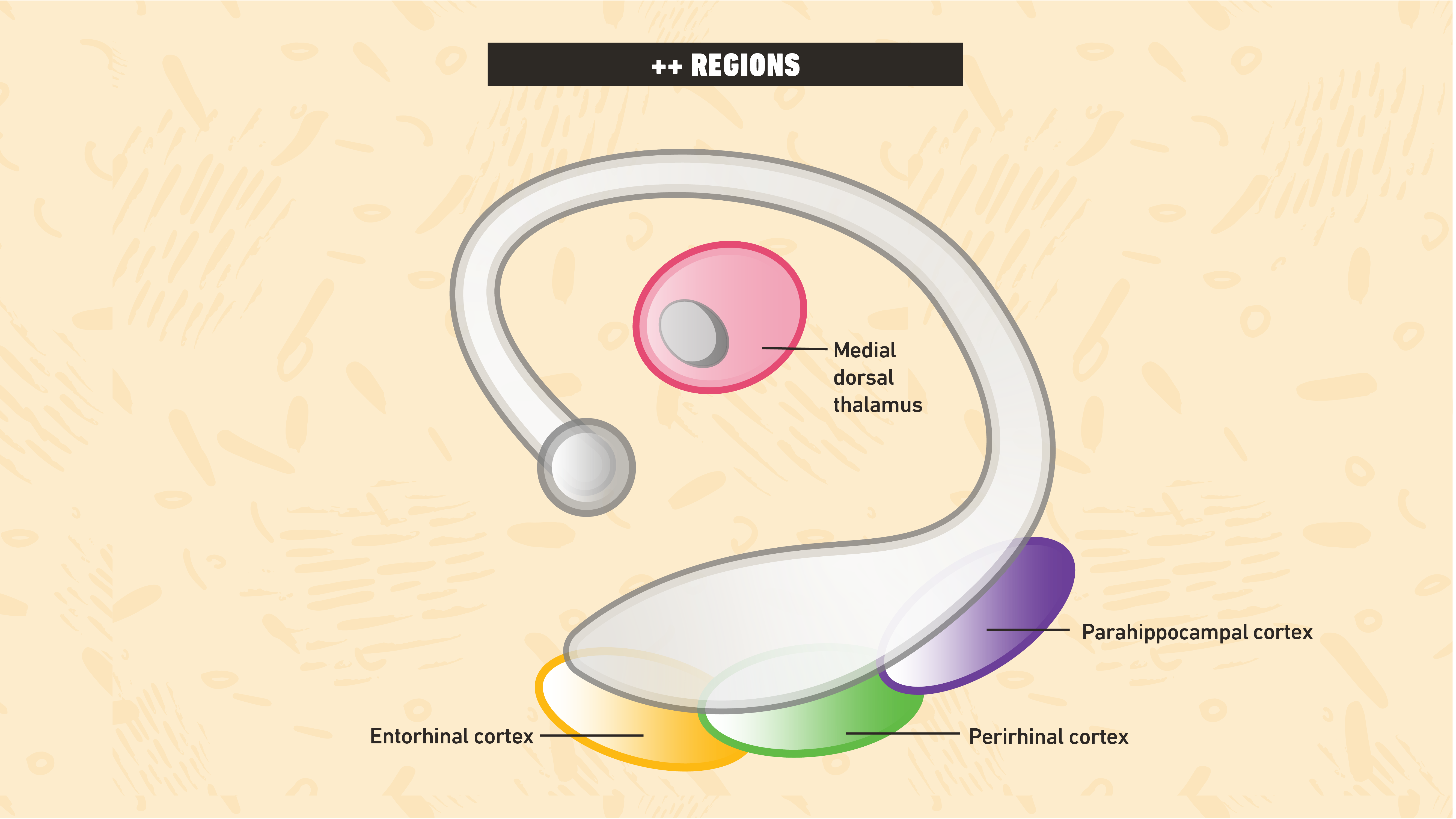
Difficulties with dissociations
So, why has it been so difficult to determine which model better accounts for amnesia? One of the primary difficulties relates to determining whether damage to one area leads to a specific impairment. For example, two famous case studies, YR and RB, suffered hippocampal damage and displayed impairments in the ability to recollect events but could still identify objects as familiar3, 4. It has been shown in other studies that the extent of damage correlates with the severity of recollection, but not familiarity, impairment5. This supports the assumption of dissociative models that the EHS supports episodic memory.
However, associative models contend that recollection and familiarity lie on a continuum, with recollection being more cognitively ‘difficult’ than familiarity6. Therefore, associative models account for such data by suggesting that this damage impaired the more challenging aspects of declarative memory, namely recollection, but that ‘easier’ forms of declarative memory, such as familiarity, are preserved. These difficulties underline why studies relying on single dissociations fail to provide definitive evidence for either associative or dissociative models.
Some studies have used double dissociations to distinguish between the models. A double dissociation refers to demonstrating that damage to ‘area A’ impairs ‘ability X’ but not ‘ability Y’, whereas damage to ‘area B’ impairs ‘ability Y’ but not ‘ability X’. Two key patients lend evidence that episodic and semantic memory represent distinct memory systems, which separate brain regions are responsible for. Patient Jon exhibited episodic memory impairment after hippocampal damage at four years of age but had relatively preserved semantic memory, such as learning word definitions7. In contrast, patient CL displayed impaired semantic memory but relatively preserved episodic memory, such as recounting autobiographical events8. These results seem to indicate that episodic and semantic memory represent distinct neurological systems.
Unfortunately, numerous limitations restrict our ability to draw conclusions from these patients. Patient CL was dyslexic, which impacts one’s ability to learn through reading and writing. This may have limited her ability to acquire and express semantic, but not episodic, memory, rather than such deficits being a consequence of neurological damage. Moreover, young children exhibit high neural plasticity. It is plausible that Jon developed compensatory mechanisms to facilitate semantic memory acquisition9. Thus, it is evident that even double dissociations struggle to distinguish between associative and dissociative models of amnesia.
Analogous amnesias?
One might think the answer to this problem is simple – if you damage the areas in the EHS that lie beyond the MTLMS and amnesia ensues, then Squire and Zola-Morgan’s (1991) associative model is incorrect. Indeed, recollection, but not familiarity10, is impaired with damage to such areas11, 12, 13. Unfortunately, solving the problem is not this simple. Associative models contend that, whilst damage to areas outside of the MTLMS can manifest a certain type of amnesia, it is distinct from MTL amnesia6. This presents a new difficulty, as one must now demonstrate that MTL amnesia is the same as amnesia arising from damage to other regions, before concluding that these studies refute associative models of amnesia.
Limitations with localization
Identifying the extent of lesions represents another major difficulty. Research is often based on patients with rather crude surgical resections of MTL structures to treat epilepsy14 or those with ischemic lesions4. Failure to accurately identify the areas that are damaged makes it much harder to find the source of impaired cognitive capacity. For example, ischemic lesions to the CA1 and CA2 subregions of the hippocampus in monkeys produce greater memory impairment than surgical removal of the entire hippocampus15, indicating that undetectable damage extended beyond the hippocampus16. Furthermore, resection of MTL structures in individuals with epilepsy necessitates damage beyond the MTL, due to its central location within the brain. Indeed, it is often unclear whether the preserved tissue still functions after surgery. For example, Rempel-Clower et al (1996) identified a group of patients with hippocampal damage that sustained nearly total hippocampal cell death, despite neuroimaging scans indicating that over 50% of the hippocampus was preserved17. Thus, identifying the extent of damage represents a fundamental difficulty in understanding the neural basis of amnesia.
In addition to identifying the locus of damage, the selectivity of lesions reflects another major challenge. The hippocampus and ++ regions are physically very close. Consequently, neurological damage often affects both sets of structures18, limiting the availability of double dissociations through selective ++ damage19. Nevertheless, sparse evidence indicates that these systems are dissociable. Patient NB suffered perirhinal cortex damage, but had a largely preserved hippocampus, and displayed profoundly impaired familiarity, with relatively preserved recollection20. These results seem to provide the crucial evidence for dissociative models, demonstrating that familiarity can be selectively impaired. However, NB’s damage extended beyond the ++ regions. Thus, despite evidence that familiarity can be selectively impaired, the rarity and complexity of such cases means we cannot definitively refute associative models of amnesia.
Turning to animal studies
Some psychologists have turned to studying animals to examine the neural basis of amnesia. However, this comes with a fundamental caveat – we do not know whether animals possess episodic memory. Demonstrating similar memory content in animals would be advantageous in studying the neural basis of amnesia, but it is very difficult to prove that animals are conscious. Nevertheless, there is some evidence in animals of ‘episodic-like’ memory (also called ‘what-where-when’ memory, which entails knowledge of what happened, where it happened and when it happened). Clayton and Dickinson (1998) have demonstrated episodic-like memory in scrub jays21. The authors reported that these birds would preferentially navigate to a location where a preferred food (worms) was stored after a short period of time, but that they would navigate to a location where a less appetizing but less degradable food (peanuts) was stored if a significant amount of time had elapsed. These rather profound findings indicate that scrub jays possess what-where-when memory.
However, there has been some controversy over whether episodic memory requires knowledge of ‘when’ an event occurred. Whilst animals (including humans) can remember how long ago an event took place, they often fail to recall the exact time it took place22, 23. Instead, we tend to use contextual knowledge to inform ‘when’ memory. This suggests that episodic memory is better defined as ‘what-where-which’ memory, entailing knowledge for what happened, where it happened and on which occasion24. Evidence for what-where-which memory in animals comes from Eacott and Norman (2004), who familiarized rats with numerous objects (what memory) at various locations (where memory) within different contexts (which memory)25. Despite each of these facets being equally familiar individually, they found that the rats preferentially navigated to novel configurations of objects at locations within contexts, supporting the notion that animals possess what-where-which memory.
In line with research on humans, the EHS of animals is also implicated in episodic-like memory. There is evidence that monkeys’ ability to recall rewarded configurations of objects in locations within specific contexts is disrupted by damage to regions within the EHS that lie beyond the MTLMS26, 27, 28. Crucially, EHS lesions do not impair performance on tasks which rely on familiarity29, supporting the notion that dissociable neural systems underlie episodic memory and familiarity.
There is also promising research indicating that there are dissociable systems within the MTLMS. These experiments have used spontaneous object recognition tasks that rely on rodents’ innate preference for exploring novel configurations of objects compared to familiar configurations. Norman and Eacott (2005) reported that rodents’ novelty preference for objects (“what” memory) and objects in locations (“what-where” memory) was selectively disrupted by perirhinal and postrhinal lesions, respectively, whereas fornix damage selectively impaired preference for novel configurations of objects in locations within contexts (what-where-which memory)30. These findings provide strong evidence that there are dissociable memory systems within the rodent MTLMS. Nevertheless, failure to demonstrate conscious awareness in animals limits the generalizability of such conclusions to humans.
Concluding comments
Determining the neurobiological basis of amnesia remains a challenge for psychologists. Much of the difficulty in settling this debate concerns limitations with our methodology. Researchers encounter difficulties with studies relying on dissociations, problems with determining whether amnesic phenotypes are analogous and limitations when attempting to localize impairment. Even studies designed with the upmost attention to detail in animals fall short of elucidating the neural underpinnings of amnesia. For now, the nature of amnesia remains somewhat elusive and whether associative or dissociative models better account for amnesia will continue to be a hotly debated topic for years to come.



