Amyloid Precursor Protein: Master or Accomplice in Alzheimer's?
Complete the form below to unlock access to ALL audio articles.
Worldwide, 50 million people are living with Alzheimer’s disease and other dementias. According to the Alzheimer’s Association, every 65 seconds someone in the United States develops this disease, which causes problems with memory, thinking and behavior.
It has been more than 100 years since Alois Alzheimer, M.D., a German psychiatrist and neuropathologist, first reported the presence of senile plaques in an Alzheimer’s disease patient brain. It led to the discovery of amyloid precursor protein that produces deposits or plaques of amyloid fragments in the brain, the suspected culprit of Alzheimer’s disease. Since then, amyloid precursor protein has been extensively studied because of its association with Alzheimer’s disease. However, amyloid precursor protein distribution within and on neurons and its function in these cells remain unclear.
A team of neuroscientists led by Florida Atlantic University’s Brain Institute sought to answer a fundamental question in their quest to combat Alzheimer’s disease — “Is amyloid precursor protein the mastermind behind Alzheimer’s disease or is it just an accomplice?”
Mutations found in amyloid precursor protein have been linked to rare cases of familial Alzheimer’s disease. Although scientists have gained a lot knowledge about how this protein turns into amyloid plaques, little is known about its native function in neurons. In the case of more common sporadic Alzheimer’s disease, the highest genetic risk factor is a protein that is involved in cholesterol transportation and not this amyloid precursor protein. Moreover, various clinical trials designed to address Alzheimer’s disease by minimizing amyloid plaque formation have failed, including one from Biogen announced last month.
In a study published in the journal Neurobiology of Disease , Qi Zhang, Ph.D., senior author, an investigator at the FAU Brain Institute, and an assistant research professor in FAU’s Schmidt College of Medicine, along with collaborators from Vanderbilt University, tackle this Alzheimer’s disease mystery by devising a multi-functional reporter for amyloid precursor protein and tracking the protein’s localization and mobility using quantitative imaging with unprecedented accuracy.
For the study, Zhang and collaborators genetically disrupted the interaction between cholesterol and amyloid precursor protein. Surprisingly, by disengaging the two, they discovered that this manipulation not only disrupts the trafficking of amyloid precursor protein but also messes up cholesterol distribution at the neuronal surface. Neurons with an altered distribution of cholesterol exhibited swollen synapses and fragmented axons and other early signs of neurodegeneration.
“Our study is intriguing because we noticed a peculiar association between amyloid precursor protein and cholesterol that resides in the cell membrane of synapses, which are points of contact among neurons and the biological basis for learning and memory,” said Zhang. “Amyloid precursor protein may just be one of the many accomplices partially contributing to cholesterol deficiency. Strangely, the heart and brain seem to meet again in the fight against bad cholesterol.”
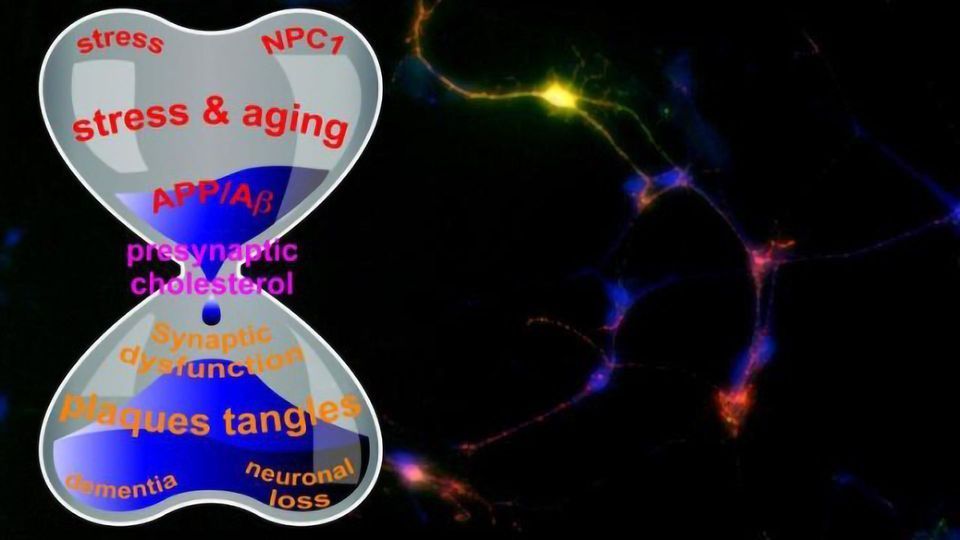
Given the broad involvement of cholesterol in almost all aspects of neurons’ life, Zhang and collaborators have proposed a new theory about the amyloid precursor protein connection in Alzheimer’s disease, especially in the surface of those tiny synapses, which triggers neurodegeneration. In this theory, a variety of pathological contributors like aging and amyloid converge on presynaptic cholesterol and the disruption of cholesterol homeostasis diverges to various pathological outcomes like synaptic dysfunction and neuronal loss.
“Although still in early stages, this cutting-edge research by Dr. Zhang and his collaborators at Vanderbilt University may have implications for the millions of people at risk for or suffering with Alzheimer’s disease,” said Randy D. Blakely, Ph.D., executive director of the FAU Brain Institute and a professor of biomedical science in FAU’s Schmidt College of Medicine. “The number of people in Florida alone who are age 65 and older with Alzheimer’s disease is expected to increase 41.2 percent by 2025 to a projected 720,000, highlighting the urgency of finding a medical breakthrough.”
This article has been republished from materials provided by Florida Atlantic University. Note: material may have been edited for length and content. For further information, please contact the cited source.
Reference: DelBove, C. E., Strothman, C. E., Lazarenko, R. M., Huang, H., Sanders, C. R., & Zhang, Q. (2019). Reciprocal modulation between amyloid precursor protein and synaptic membrane cholesterol revealed by live cell imaging. Neurobiology of Disease, 127, 449–461. https://doi.org/10.1016/j.nbd.2019.03.009

