New insights into underlying cellular mechanisms of information processing in the brain

Complete the form below to unlock access to ALL audio articles.
Researchers have uncovered a key factor in regulating information transmittal during the early stages of auditory processing
The human body consists of almost 100 billion neurons that contain synapses. Every function, feeling or action depends on the ability of synapses to maintain constant communication between neurons, transmitting information via continuous release of neurotransmitters from synaptic vesicles. A new study has discovered that a key factor in regulating this continual communication is the proximity of synaptic vesicles (SVs) next to voltage gated calcium channels within synapses.
Synaptic vesicles in cell-to-cell communication
While synapses contain hundreds to thousands of synaptic vesicles, when a specific signal is received by the neuron, only a fraction of synaptic vesicles that exist in what is called the readily releasable pool are discharged from one neuron to another. Previous studies have demonstrated that within the readily releasable pool, synaptic vesicles have different specific kinetic properties of release that impact the type and bandwidth of information that can be relayed in response to stimulation. What was not known was the cellular mechanisms that regulate the release of synaptic vesicles from the readily releasable pool to support the early stages of auditory processing.
New findings
Published in the February edition of the Journal of Neuroscience, the authors of the manuscript report that a dominant factor regulating the properties of synaptic vesicle release supporting the early stages of auditory processing is the distance between the synaptic vesicles and voltage gated calcium channels within a synapse. The authors characterized the readily releasable pool of synaptic vesicles at the calyx of Held, a critical component of auditory processing, employing several different techniques that allowed them to investigate the pre-synaptic mechanisms of informational transmittal. This study identifies the critical mechanism is the regulation of Ca2+ influx through voltage gated calcium channels and release of neurotransmitters by synaptic vesicles. These findings are important for understanding the mechanisms of synaptic transmission, specifically for neuronal circuits that rely on fast, continuous synaptic transmission.
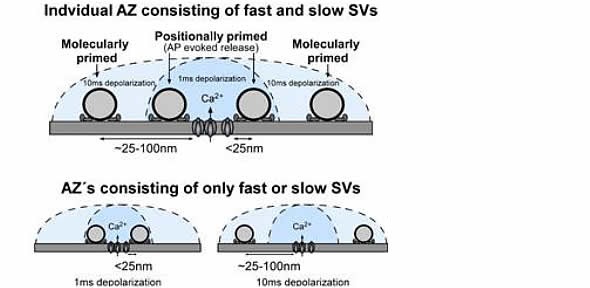
SV distribution within the P16 –P19 calyx RRP. Schematic diagram showing SVs at different priming states located at different distance to the Ca 2+ source. Only the SVs that locate close to the Ca 2+ source can be released by AP. We propose that either a single AZ contains a mixture of fast and slow SVs or that there are individual AZs within the calyx that contain only fast or only slow pool SVs. In both cases, we propose that distal SVs are rapidly converted to fast pool SVs for AP-evoked release to maintain signaling at high firing rates. Credit: Max Planck Florida Institute for Neuroscience; Dr. Samuel M. Young, Jr.
“It is becoming apparent that the underlying cause of most neuropsychiatric or neurodegenerative diseases is a dysfunction of the synapse,” explained Dr. Young. “Identifying what factors are involved in proper synaptic transmission and neural circuit function have tremendous potential as therapies for neurological disorders or brain injury.”
Future directions
According to Dr. Young, the future goals of this project are to uncover molecular mechanisms that allow synapses to sustain synaptic transmission over a wide range of activity levels to allow for proper information processing by the neuronal circuit in which they are embedded.
Note: Material may have been edited for length and content. For further information, please contact the cited source.
Max Planck Florida Institute for Neuroscience press release
Publication
Zuxin Chen, Brati Das, Yukihiro Nakamura, David A. DiGregorio, Samuel M. Young Jr. Ca2+ Channel to Synaptic Vesicle Distance Accounts for the Readily Releasable Pool Kinetics at a Functionally Mature Auditory Synapse . Journal of Neuroscience, Published February 4 2015. doi: 10.1523/JNEUROSCI.2753-14.2015

